|
From there, the absorbance will just depend on the concentration of the permanganate.Tris(bipyridine)ruthenium(II) chloride is the chloride salt coordination complex with the formula 2+ 2Cl −. Most cuvettes are 1 cm wide if the extinction coefficient contains a unit of cm -1, we can just use b = 1 cm and that part is taken care of. In which \(ε\) = extinction coefficient, \(l\) = path length, \(c\) = concentration. 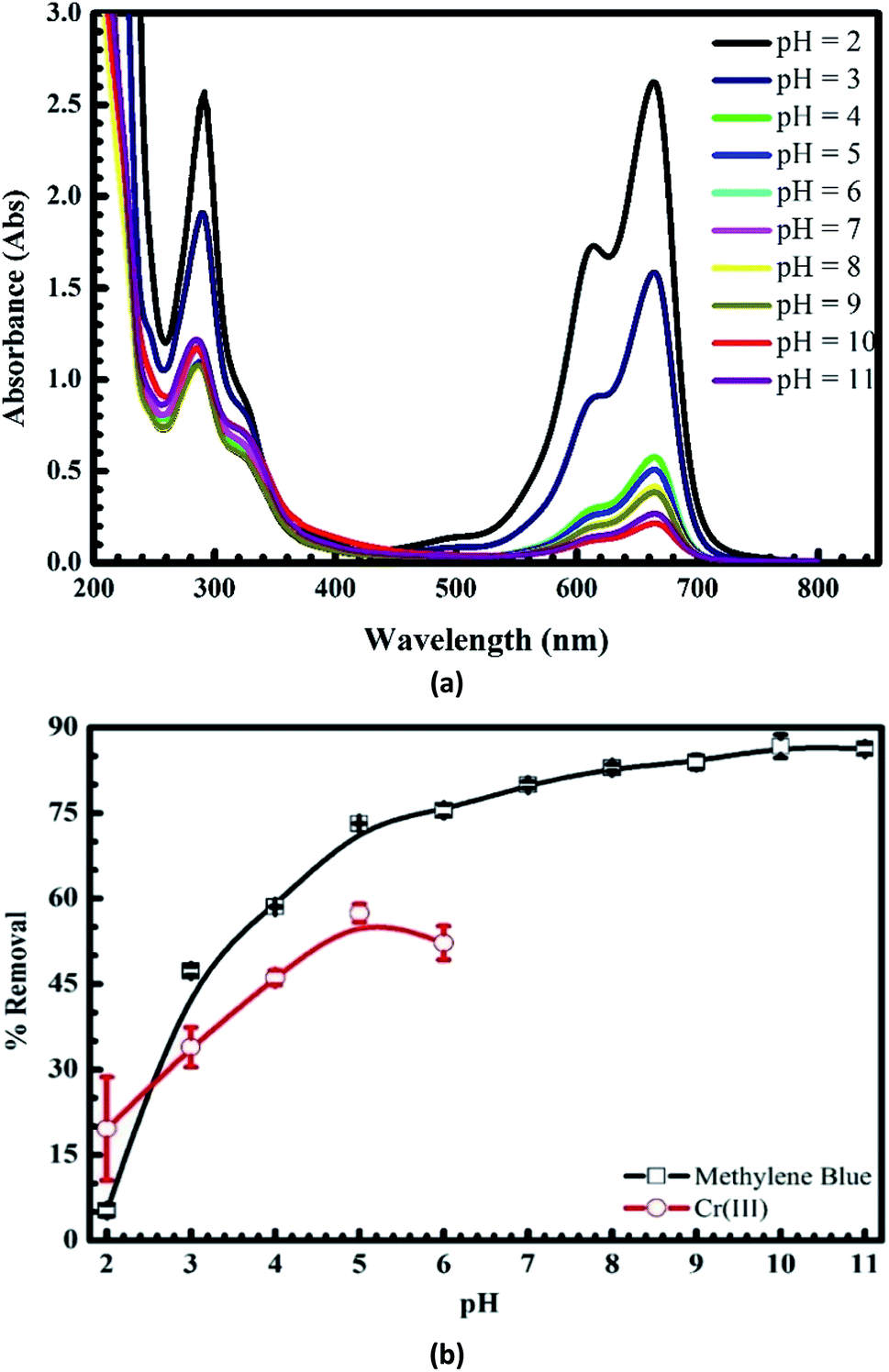 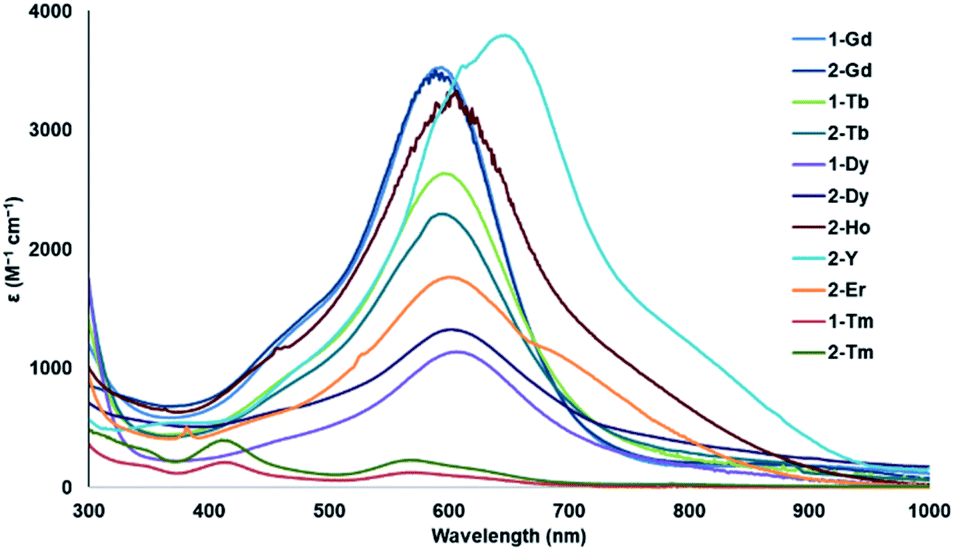
Also, the longer the light travels through the cuvette, the more permanganate it will encounter, and so the more light it will absorb. First of all, how concentrated is the sample? The more potassium permanganate there is dissolved in the sample, the more light it can absorb. How much light is absorbed by a sample of potassium permanganate at that wavelength depends on a couple of additional factors, however. How strongly a compound absorbs photons at a particular wavelength is described by a quantity called the extinction coefficient (or alternatively, in different variations, the absorption coefficient or the molar absorptivity coefficient).įor example, it can be shown that aqueous potassium permanganate, KMnO 4, at its absorption maximum of 530 nm, has an extinction coefficient ε = 14 L g -1 cm -1. Knowing that information might help us distinguish the spectrum of that compound from the spectrum of another that has similar absorption wavelengths, but that shows peaks in the UV spectrum of very different sizes. A specific compound might absorb at 250 nm and at 450 nm however, an additional characteristic of that compound might be that it absorbs very strongly at 250 nm and only weakly at 450 nm. Now we see that the y axis also matters to some extent. We might be able to look at the UV spectrum of a compound and tell its identity or structure that task would be especially straightforward if we had a few different options to choose from. The whole idea of UV spectroscopy is that different compounds might absorb photons of different wavelengths based on their electronic structures. In the preceding spectra, predict the observed color of each of the complexes. We just need to understand that these distinctions can influence what UV spectra look like. We aren't going to worry about why different electronic transitions occur with greater or lesser efficiency that has to do with symmetry and group theory, and some mathematics that we aren't equipped to handle right now. MLCT transitions require much more energy but they happen frequently, leading to stronger absorbances in the spectrum. These electronic transitions interact with photons very efficiently.Īs a result, there is a kind of counterintuitive relationship in the UV-visible spectra of transition metal complexes: d-d transitions require very little energy but occur relatively infrequently, meaning they give very weak absorbances in the spectrum. The electronic transitions involved just are not very good at capturing photons.īy comparison, it might sound like it would be difficult to move an electron from the metal all the way to the ligand, but it's actually pretty easy. That sounds like it might be pretty easy - the electron isn't going very far, after all - but d-d transitions are actually quite inefficient. For transition metals, the valence electrons are in the d sub-shell, and in a d-d transition, the electron is excited from one d level to another. For transition metals, these electronic excitations are called d-d transitions. In addition, sometimes electrons can be excited from one level to another, just on the metal ion. However, these two acronyms are sometimes used interchageably to suggest some sort of transition that involves both the ligand and the metal, without worrying too much about the direction. Sometimes the former case is referred to as a ligand-to-metal charge transfer, or LMCT the latter case would be a metal-to-ligand charge transfer, or MLCT. It involves the excitation of an electron from the ligand to the metal, or vice versa. One very common transition is called a charge transfer transition. These transitions might involve the metal ion itself, or the ligands - those molecules or ions that bind to the metal ion.  
When they do, there are a number of possible electronic transitions that can result. These transition metal complexes or coordination complexes have lots of electrons, and they can often interact with lots of different photons. These molecules bind to the metal ions, forming coordination complexes. In solution, metal ions would not swim around by themselves, but would attract other molecules to them. Because of their relatively low electronegativity, transition metals are frequently found as positively-charged ions, or cations. Transition metals are often associated with brightly-colored compounds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
